  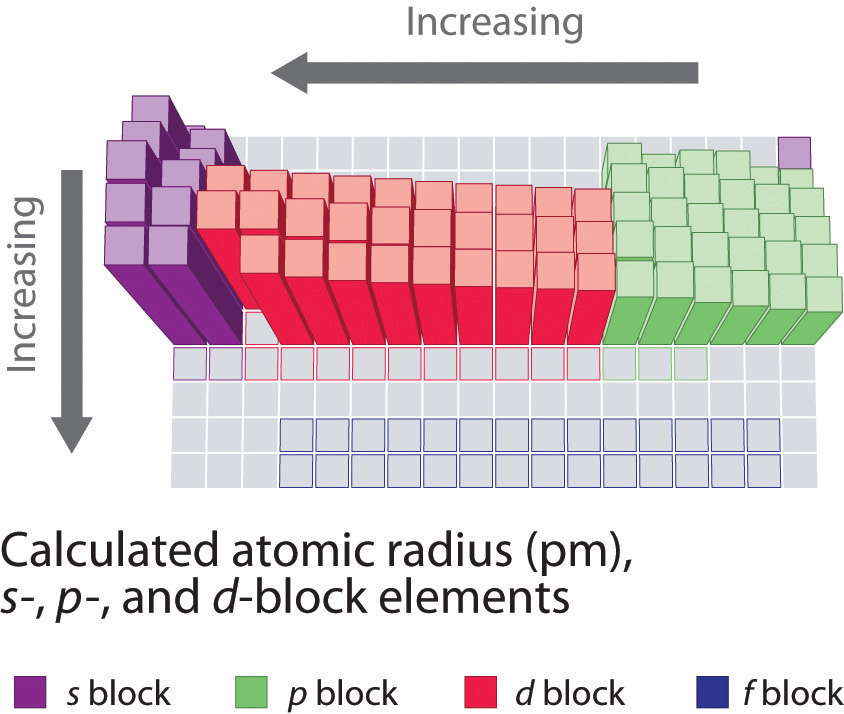
Similarly, as we proceed across the row, the increasing nuclear charge is not effectively neutralized by the electrons being added to the s and p orbitals. Consequently, beryllium is significantly smaller than lithium. This means that the effective nuclear charge experienced by the s electrons in beryllium is between +1 and +2 (the calculated value is +1.66). Electron density diminishes gradually with increasing distance, which makes it impossible to draw a sharp line marking the boundary of an atom.įigure 1: Plots of Radial Probability as a Function of Distance from the Nucleus for \ce.) In contrast, the two s electrons in beryllium do not shield each other very well, although the filled s 2 shell effectively neutralizes two of the four positive charges in the nucleus. This point is illustrated in Figure 4.3.1 which shows a plot of total electron density for all occupied orbitals for three noble gases as a function of their distance from the nucleus. Recall that the probability of finding an electron in the various available orbitals falls off slowly as the distance from the nucleus increases. In this chapter, we will discuss how atomic and ion “sizes” are defined and obtained. As a result, atoms and ions cannot be said to have exact sizes however, some atoms are larger or smaller than others, and this influences their chemistry. To understand periodic trends in atomic radii.Īlthough some people fall into the trap of visualizing atoms and ions as small, hard spheres similar to miniature table-tennis balls or marbles, the quantum mechanical model tells us that their shapes and boundaries are much less definite than those images suggest.From the table above, a single bond between fluorine and nitrogen has a bond length of approximately 64 + 71 =135 pm. The bond between fluorine and nitrogen is a single bond. To find the nitrogen-to-fluorine bond length in NF 3, draw the Lewis structure. Therefore, the bond length is greater in CO 2.ĥ. Therefore, the bond length is greater in CO 2.Īnother method makes use of the fact that the more electron bonds between the atoms, the tighter the electrons are pulling the atoms together. Referring to the table above, a double bond between carbon and oxygen has a bond length of approximately 67 + 57 = 124 pm and a triple bond between carbon and oxygen has a bond length of approximately 60 + 53 =113 pm. From the Lewis structures for CO 2 and CO, there is a double bond between the carbon and oxygen in CO 2 and a triple bond between the carbon and oxygen in CO. The bond between carbon and nitrogen is a triple bond, and a triple bond between carbon and nitrogen has a bond length of approximately 60 + 54 =114 pm.Ĥ. 
To find the carbon-nitrogen bond length in HCN, draw the Lewis structure of HCN. Adding these together and dividing by the number of bonds (3) reveals that the bond order of nitrate is 1.33.ģ. N=O has a bond order of two, and both N-O bonds have a bond order of one. To find the bond order of this molecule, take the average of the bond orders. The Lewis structure for NO 3 - is given below: There is a double bond between the two oxygen atoms therefore, the bond order of the molecule is 2.Ģ. First, write the Lewis structure for \(O_2\). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
